Top Previous Year Questions - Atomic Physics
Question
A light of energy is incident on a hydrogen atom in its ground state. The atom absorbs the radiation and reaches to one of its excited states. The angular momentum of the atom in the excited state is . The value of is ______ (use )
JEE Main 2023 (01 Feb Shift 1)
Enter your answer
Explaination
Question
Which of the following figure represents the variation of with (if radius of a nucleus and its mass number)
JEE Main 2022 (25 Jun Shift 2)
Options
-
A:
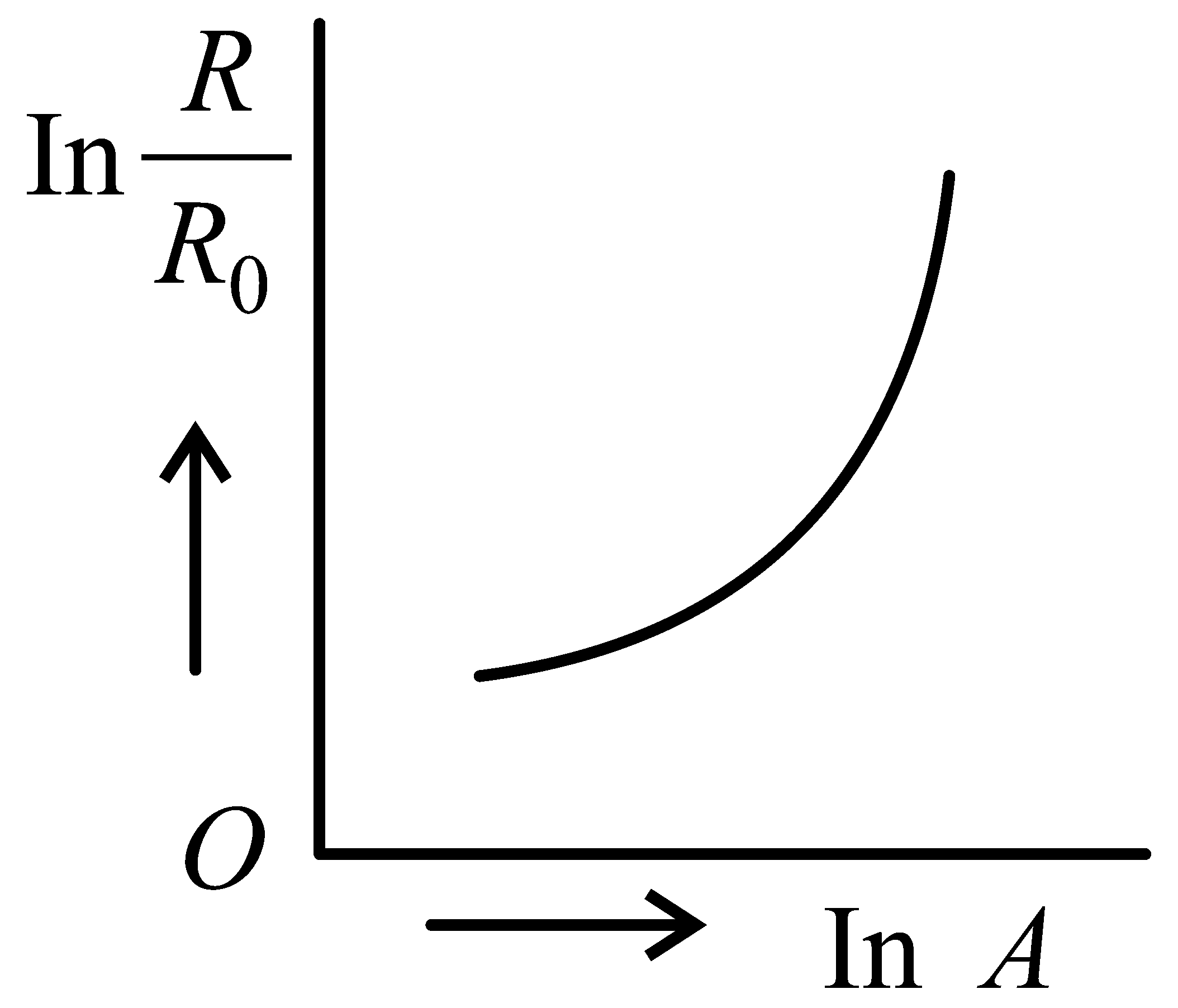
-
B:
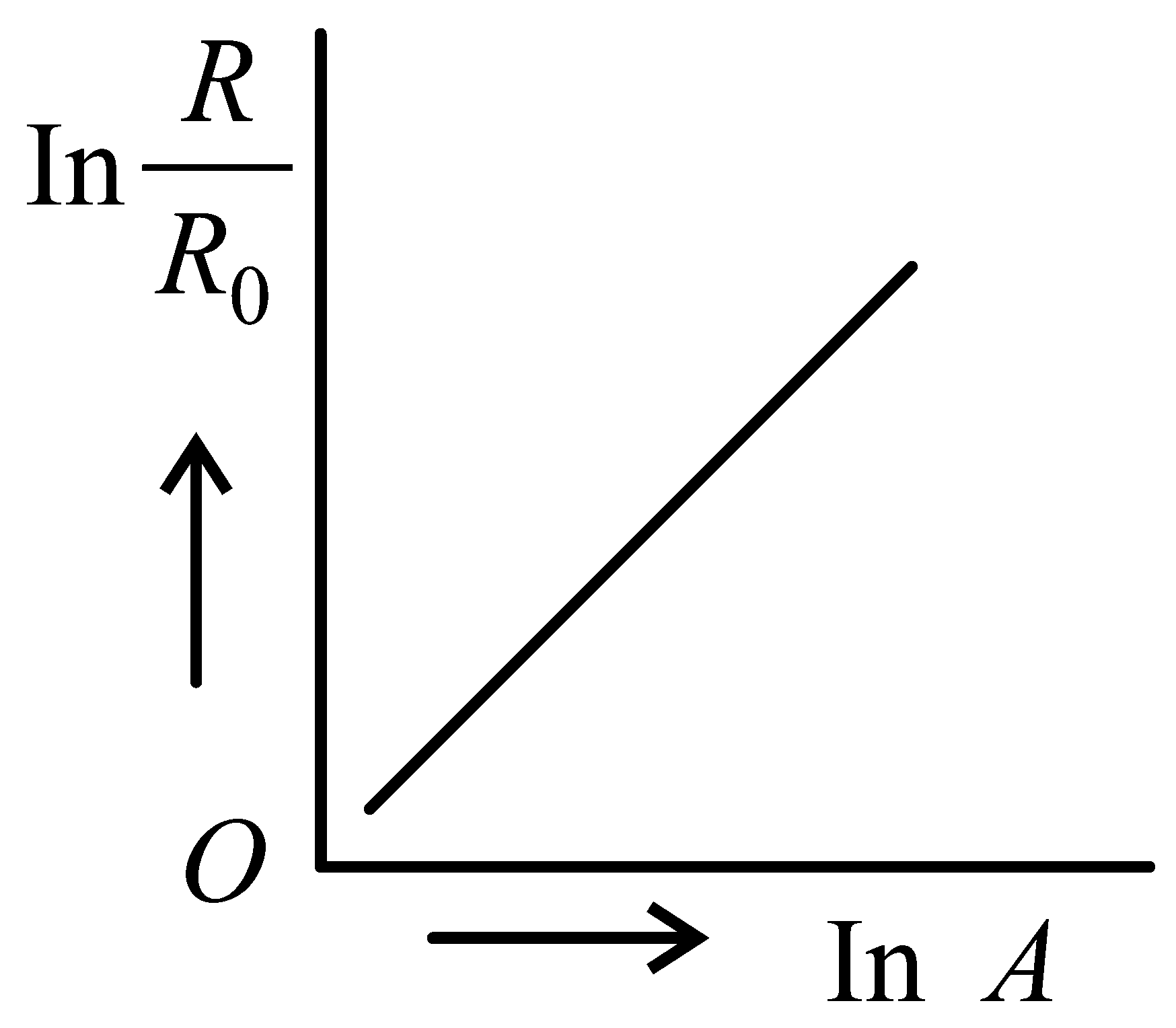
-
C:
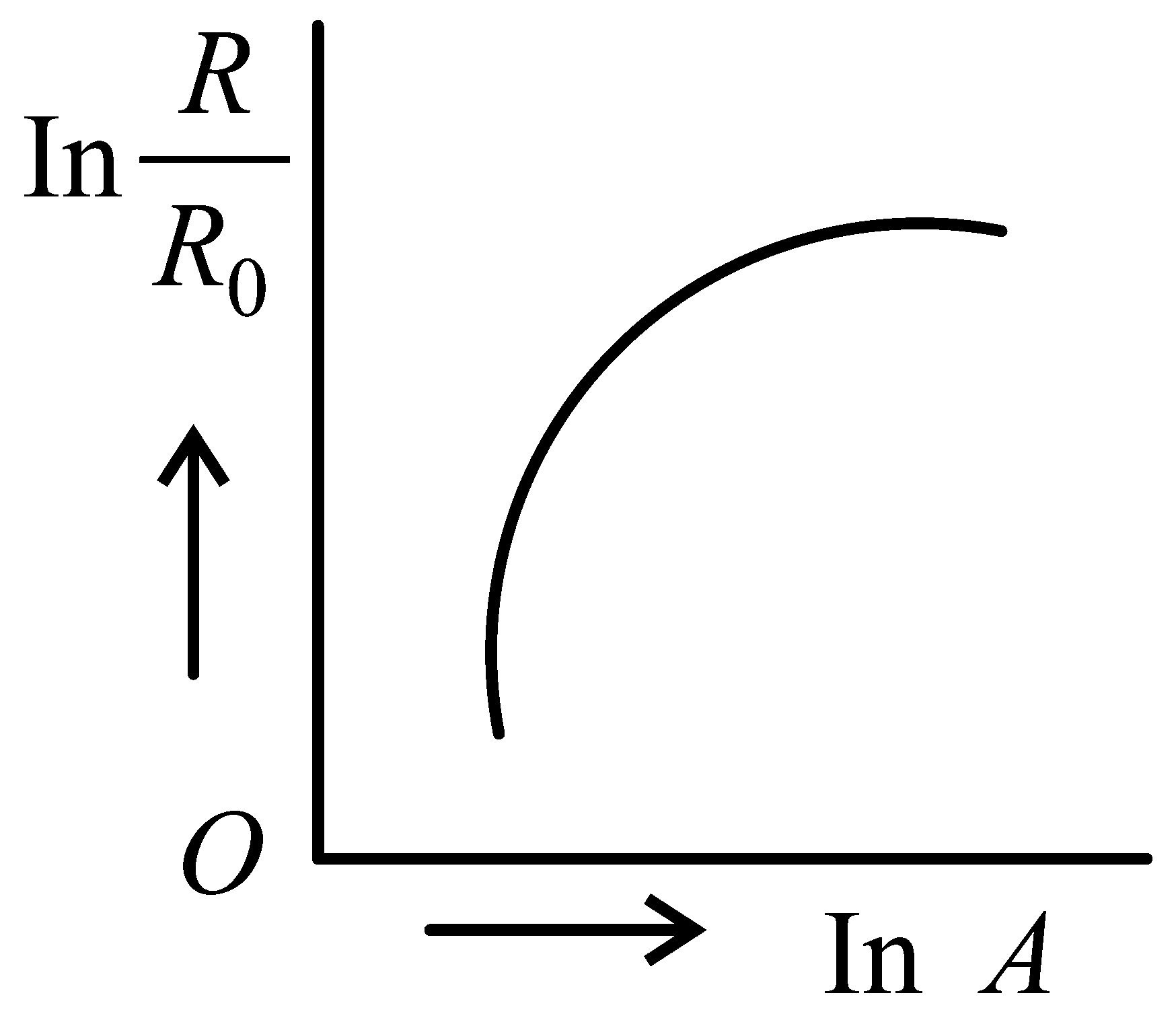
-
D:
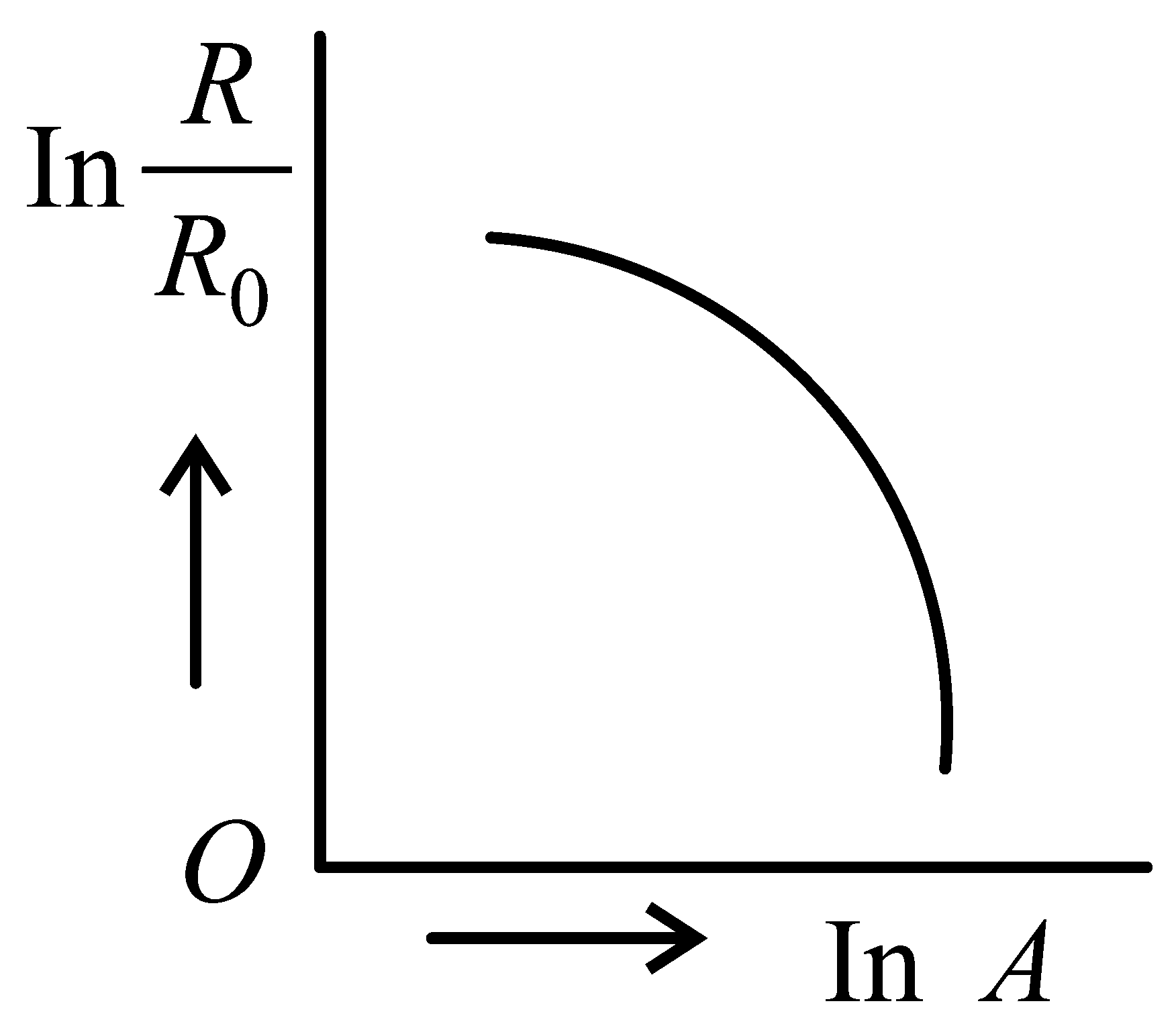
Explaination
Question
Find the ratio of energies of photons produced due to transition of an election of hydrogen atom from its(i) second permitted energy level to the first level, and (ii) the highest permitted energy level to the first permitted level.
JEE Main 2022 (29 Jul Shift 1)
Options
- A:
- B:
- C:
- D:
Explaination
Question
A hydrogen atom changes its state from $n=3$ to $n=2$. Due to recoil, the percentage change in the wave length of emitted light is approximately $1 \times 10^{-n}$. The value of $n$ is_____. [Given $\mathrm{Rhc}=13.6 \mathrm{eV}, \mathrm{hc}=1242 \mathrm{eV} \mathrm{nm}, \mathrm{h}=6.6 \times 10^{-34} \mathrm{~J} \mathrm{~s}$ mass of the hydrogenatom $=1.6 \times 10^{-27} \mathrm{~kg}$ ]
JEE Main 2024 (04 Apr Shift 1)
Enter your answer
Explaination
Question
The first three spectral lines of -atom in the Balmer series are given considering the Bohr atomic model, the wave lengths of first and third spectral lines are related by a factor of approximately . The value of , to the nearest integer, is ________.
JEE Main 2021 (16 Mar Shift 1)
Enter your answer
Explaination
Question
In an alpha particle scattering experiment distance of closest approach for the $\alpha$ particle is $4.5 \times 10^{-14} \mathrm{~m}$. If target nucleus has atomic number 80 , then maximum velocity of $\alpha$ - particle is _______ $\times 10^5 \mathrm{~m} / \mathrm{s}$ approximately. $\left(\frac{1}{4 \pi \epsilon_0}=9 \times 10^9\right.$ SI unit, mass of $\alpha$ particle $\left.=6.72 \times 10^{-27} \mathrm{~kg}\right)$
JEE Main 2024 (08 Apr Shift 1)
Enter your answer
Explaination
Question
In the given figure, the energy levels of hydrogen atom have been shown along with some transitions marked and .
The transitions and respectively represent
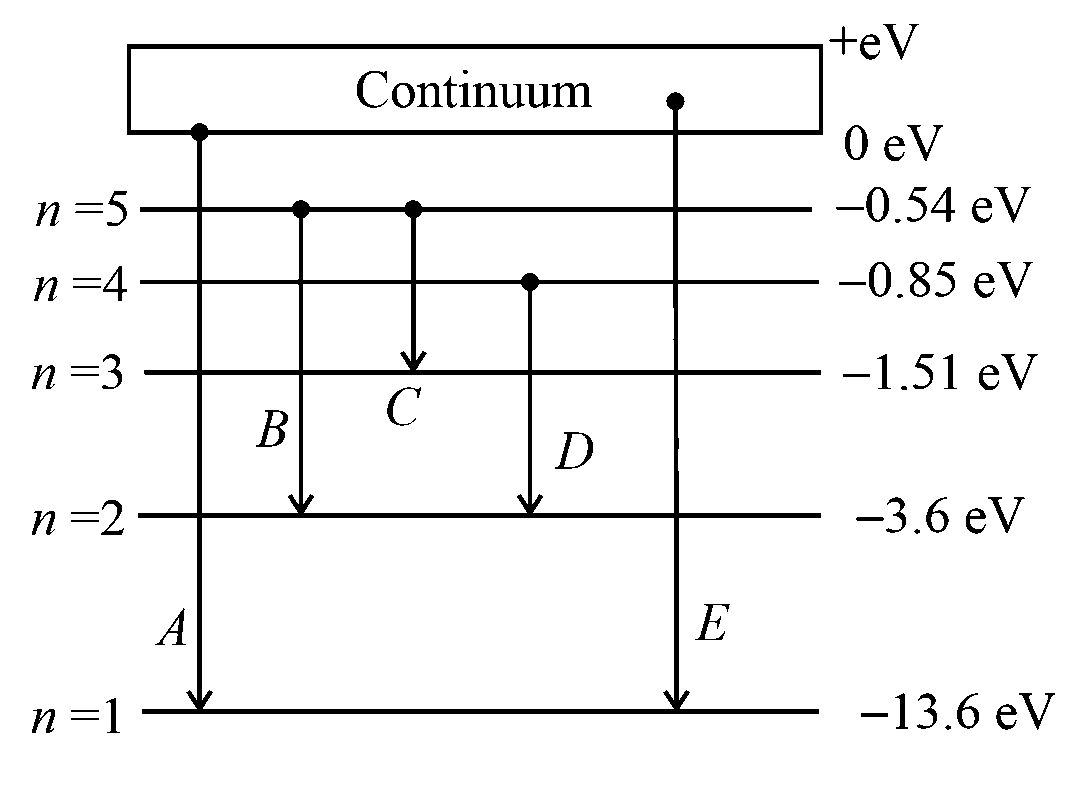
JEE Main 2021 (24 Feb Shift 1)
Options
- A: The ionization potential of hydrogen, second member of Balmer series and third member of Paschen series.
- B: The series limit of Lyman series, second member of Balmer series and second member of Paschen series.
- C: The series limit of Lyman series, third member of Balmer series and second member of Paschen series.
- D: The first member of the Lyman series, third member of Balmer series and second member of Paschen series.
Explaination
Question
Hydrogen atom is bombarded with electrons accelerated through a potential different of , which causes excitation of hydrogen atoms. If the experiment is being formed at . The minimum potential difference needed to observe any Balmer series lines in the emission spectra will be , where _________.(Write the value to the nearest integer)
JEE Main 2024 (29 Jan Shift 2)
Enter your answer
Explaination
Question
A particular hydrogen - like ion emits the radiation of frequency when it makes transition from to The frequency of radiation emitted in transition from to is when _____.
JEE Main 2024 (01 Feb Shift 2)
Enter your answer
Explaination
Question
The first member of the Balmer series of hydrogen atom has a wavelength of . The wavelength of the second member of the Balmer series (in nm) is_____________
JEE Main 2020 (08 Jan Shift 2)
Enter your answer
Explaination
Question
The ratio of the shortest wavelength of Balmer series to the shortest wavelength of Lyman series for hydrogen atom is :
JEE Main 2024 (06 Apr Shift 1)
Options
- A: $4: 1$
- B: $1: 4$
- C: $2: 1$
- D: $1: 2$
Explaination
Question
Given below are two statements
Statement I: In hydrogen atom, the frequency of radiation emitted when an electron jumps from lower energy orbit to higher energy orbit , is given as
Statement II: The jumping of electron from higher energy orbit to lower energy orbit is associated with frequency of radiation given as . This condition is Bohr's frequency condition.
In the light of the above statements, choose the correct answer from the options given below:
JEE Main 2022 (27 Jun Shift 2)
Options
- A: Both statement I and statement II are true.
- B: Both statement I and statement II are false.
- C: Statement I is correct but statement II is false.
- D: Statement I is incorrect but statement II is true.
Explaination
Question
In Bohr's atomic model, the electron is assumed to revolve in a circular orbit of radius If the speed of electron is Then the current associated with the electron will be__________ [Take as
JEE Main 2021 (27 Jul Shift 1)
Enter your answer
Explaination
Question
is the ratio of energies of photons produced due to transition of an electron of hydrogen atom from its
(i) third permitted energy level to the second level and
(ii) the highest permitted energy level to the second permitted level.
The value of will be
JEE Main 2022 (25 Jul Shift 2)
Enter your answer
Explaination
Question
The graph which depicts the results of Rutherford gold foil experiment with -particles is:
Scattering angle
Number of scattered -particles detected
(Plots are schematic and not to scale)
JEE Main 2020 (08 Jan Shift 1)
Options
-
A:
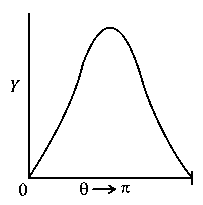
-
B:
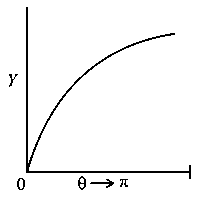
-
C:
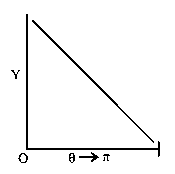
-
D:
Explaination
Question
In Bohr's atomic model of hydrogen, let and are the kinetic energy, potential energy and total energy of the electron respectively. Choose the correct option when the electron undergoes transitions to a higher level :
JEE Main 2022 (24 Jun Shift 2)
Options
- A: All and increase.
- B: decreases, and increase.
- C: decreases, and increase.
- D: increases, and decrease